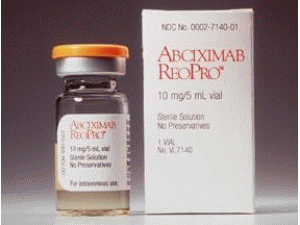
阿昔单抗注射剂abciximab(ReoPro injection 10mg/5m)
 产地国家:美国
产地国家:美国
处方药:是
所属类别: 10毫克/5毫升/瓶
包装规格: 10毫克/5毫升/瓶
计价单位:瓶
生产厂家英文名:J-O-M PHARM SERVICES
原产地英文商品名:ReoPro injection 10mg/5mL/vial
原产地英文药品名:abciximab
中文参考商品译名:ReoPro注射剂 10毫克/5毫升/瓶
中文参考药品译名:阿昔单抗
简介
部份中文阿昔单抗处方资料(仅供参考)
商品名:ReoPro。
英文名:abciximab。
中文名:阿昔单抗注射液。
生产商:J-O-M PHARM SERVICES
药理学
阿昔单抗(英文别名Centorx、Reopro)本品是一种带有精氨酸-甘氨酸-天门冬氨酸(RGD)合成的肽按顺序与血小板糖蛋白Ⅱb/Ⅲ受体结合。本品不是与RGD位点结合,而是阻断大分子进入受体(通过空间障碍和/或结构形态的作用)。本品为嵌合性单克隆抗体7E3的碎片,它与血小板表面的糖蛋白Ⅱb/Ⅲa受体结合,以阻断纤维蛋白元,VWD因子和其它有粘性的分子与受体位点结合,从而抑制血小板聚集防止形成血栓,堵塞血流而致心肌急性缺血。
药动学:静脉注射本品后,游离血小板数量迅速下降,主要发生在第一个半衰期(10min)内和第二个半衰期(30min)内,作用快可能是因为该药与血小板糖蛋白Ⅱb/Ⅲa受体结合迅速。与以每分钟10mg阿司匹林给药的患者作对照,本品静脉滴注(0.25mg/kg)2小时后,抑制了90%以上的血小板凝集。给药10天后,仍出现少量的糖蛋白Ⅱb/Ⅲa受体阻断。
适应症:
1.本品适用于经皮穿刺冠状血管成形术或动脉粥样化切除术,为防止病人突然发生冠状血管堵塞引起心肌急性缺血的辅助治疗。
2.处于突然发生堵塞的高危病人,至少要伴有以下情况之一:不稳定的心绞痛或无Q性心肌梗塞;在12小时内发作的急性Q性心肌梗塞;在扩张动脉时Ⅱ型血管损伤;至少65岁以上的妇女,在扩张动脉时Ⅰ型血管损伤;糖尿病人扩张动脉时Ⅰ型血管损伤或与7日内发生的心肌梗塞有关的血管成形术。在这些情况下,本品与阿司匹林和肝素是必须使用的。
3.本品对正在进行的血管成形术有抗血栓形成的活性并可预防血管再狭窄的发生。
用法用量:在血管成形术前10分钟,由静脉滴注本品按每公斤体重250μg,滴注1分钟以上,然后以每分钟滴入10μg,维持12小时。
给药说明:可能发生血小板减少,故给予本品前后应监测血小板。给予本品后2~4周可能产生抗体,因此,当再用本品或其它单克隆抗体后可能发生过敏反应,故不宜重复给予本品治疗。单次给予本品未发现过敏反应,但是这种可能性应当注意。以下情况需加用本品(虽会增加出血的危险):经皮穿刺冠脉血管成形术后12小时发生急性心肌梗塞;手术时间延长(70min以上);手术失败。肝素等抗凝药也会增加出血的危险。若出现严重出血,本品和合用的肝素应立即停用。
禁用慎用:给药后36小时出血是最常见的不良反应。因此,本品不应给予活动性出血或有出血倾向的病人。本品还未经过动物遗传研究,因此尚不清楚用于孕妇时会否引起致命危害或影响胎儿。除非确实需要,才能将本品用于孕妇。本品对儿童及哺乳期妇女的安全情况尚未确定,故不宜使用。由于本品会增加出血的危险,故有以下情况者禁用:急性内出血;近期内(6周内)胃肠道出血或泌尿道出血;2年内的脑意外损伤;脑损伤出现明显的神经系统缺陷;有出血倾向;7天内口服抗凝药(除非凝血时间低于对照组的1.2倍);血小板减少症(<100000 cells/ul);近期内(6周内)作过大的外科手术或有严重损伤;颅内肿瘤;动静脉畸形或动脉瘤;严重的失控性高血压;有脉管炎史;经皮透腔血管成形手术前或手术中注射了右旋糖酐者;对本品或鼠蛋白过敏者。
不良反应:给药后36小时出血是最常见的不良反应。因此,本品不应给予活动性出血或有出血倾向的病人。其它不良反应包括低血压、恶心、呕吐、头痛、心动过缓、发热和血管功能障碍等。
贮藏:本品应冷藏,但不可冻结或振摇。应储存在2至8°C(36至46°F)的温度下。 不要冻结。
英文版说明书
ReoPro (abciximab)IndicationsAbciximab is indicated as an adjunct to percutaneous coronary intervention for the prevention of cardiac ischemic complicationsin patients undergoing percutaneous coronary interventionin patients with unstable angina not responding to conventional medical therapy when percutaneous coronary intervention is planned within 24 hoursSafety and efficacy of Abciximab use in patients not undergoing percutaneous coronary intervention have not been established.Abciximab is intended for use with aspirin and heparin and has been studied only in that setting, as described in CLINICAL STUDIES.Contraindications:Because Abciximab may increase the risk of bleeding, Abciximab is contraindicated in the following clinical situations:Active internal bleedingRecent (within six weeks) gastrointestinal (GI) or genitourinary (GU) bleeding of clinical significanceHistory of cerebrovascular accident (CVA) within two years, or CVA with a significant residual neurological deficitBleeding diathesisAdministration of oral anticoagulants within seven days unless prothrombin time is ≤ 1.2 times controlThrombocytopenia (< 100,000 cells/μL)Recent (within six weeks) major surgery or traumaIntracranial neoplasm, arteriovenous malformation, or aneurysmSevere uncontrolled hypertensionPresumed or documented history of vasculitisUse of intravenous dextran before PCI, or intent to use it during an interventionAbciximab is also contraindicated in patients with known hypersensitivity to any component of this product or to murine proteins.Adverse Reactions:Bleeding- Abciximab has the potential to increase the risk of bleeding, particularly in the presence of anticoagulation, e.g., from heparin, other anticoagulants or thrombolytics. Bleeding in the Phase 3 trials was classified as major, minor or insignificant by the criteria of the Thrombolysis in Myocardial Infarction study group (16). Major bleeding events were defined as either an intracranial hemorrhage or a decrease in hemoglobin greater than 5 g/dL. Minor bleeding events included spontaneous gross hematuria, spontaneous hematemesis, observed blood loss with a hemoglobin decrease of more than 3 g/dL, or a decrease in hemoglobin of at least 4 g/dL without an identified bleeding site. Insignificant bleeding events were defined as a decrease in hemoglobin of less than 3 g/dL or a decrease in hemoglobin between 3-4 g/dL without observed bleeding. In patients who received transfusions, the number of units of blood lost was estimated through an adaptation of the method of Landefeld, et al. (17).In the EPIC trial, in which a non-weight-adjusted, longer-duration heparin dose regimen was used, the most common complication during Abciximab therapy was bleeding during the first 36 hours. The incidences of major bleeding, minor bleeding and transfusion of blood products were significantly increased. Major bleeding occurred in 10.6% of patients in the Abciximab bolus plus infusion arm compared with 3.3% of patients in the placebo arm. Minor bleeding was seen in 16.8% of Abciximab bolus plus infusion patients and 9.2% of placebo patients (7). Approximately 70% of Abciximab-treated patients with major bleeding had bleeding at the arterial access site in the groin. Abciximab-treated patients also had a higher incidence of major bleeding events from gastrointestinal, genitourinary, retroperitoneal, and other sites.Bleeding rates were reduced in the CAPTURE trial, and further reduced in the EPILOG and EPISTENT trials by use of modified dosing regimens and specific patient management techniques. In EPILOG and EPISTENT, using the heparin and Abciximab dosing, sheath removal and arterial access site guidelines described under PRECAUTIONS, the incidence of major bleeding in patients treated with Abciximab and low-dose, weight-adjusted heparin was not significantly different from that in patients receiving placebo.Subgroup analyses in the EPIC and CAPTURE trials showed that non-CABG major bleeding was more common in Abciximab patients weighing ≤ 75 kg. In the EPILOG and EPISTENT trials, which used weight-adjusted heparin dosing, the non-CABG major bleeding rates for Abciximab-treated patients did not differ substantially by weight subgroup.Although data are limited, Abciximab treatment was not associated with excess major bleeding in patients who underwent CABG surgery. (The range among all treatment arms was 3-5% in EPIC, and 1-2% in the CAPTURE, EPILOG, and EPISTENT trials.) Some patients with prolonged bleeding times received platelet transfusions to correct the bleeding time prior to surgery. (see PRECAUTIONS: Restoration of Platelet Function.)The rates of major bleeding, minor bleeding and bleeding events requiring transfusions in the CAPTURE, EPILOG, and EPISTENT trials are shown in Table 4. The rates of insignificant bleeding events are not included in Table 4.Cases of fatal bleeding have been reported rarely during post-marketing use of Abciximab (see WARNINGS: Bleeding Events).Pulmonary alveolar hemorrhage has been rarely reported during use of Abciximab. This can present with any or all of the following in close association with ReoPro administration: hypoxemia, alveolar infiltrates on chest x-ray, hemoptysis, or an unexplained drop in hemoglobin.Intracranial Hemorrhage and Stroke- The total incidence of intracranial hemorrhage and non-hemorrhagic stroke across all four trials was not significantly different, 9/3023 for placebo patients and 15/4680 for Abciximab-treated patients. The incidence of intracranial hemorrhage was 3/3023 for placebo patients and 7/4680 for Abciximab patients.Thrombocytopenia- In the clinical trials, patients treated with Abciximab were more likely than patients treated with placebo to experience decreases in platelet counts.Among patients in the EPILOG and EPISTENT trials who were treated with Abciximab plus low-dose heparin, the proportion of patients with any thrombocytopenia (platelets less than 100,000 cells/μL) ranged from 2.5 to 3.0%. The incidence of severe thrombocytopenia (platelets less than 50,000 cells/μL) ranged from 0.4 to 1.0% and platelet transfusions were required in 0.9 to 1.1%, respectively. Modestly lower rates were observed among patients treated with placebo plus standard-dose heparin. Overall higher rates were observed among patients in the EPIC and CAPTURE trials treated with Abciximab plus longer duration heparin: 2.6 to 5.2% were found to have any thrombocytopenia, 0.9 to 1.7% had severe thrombocytopenia, and 2.1 to 5.5% required platelet transfusion, respectively.In a readministration registry study of patients receiving a second or subsequent exposure to Abciximab (see PRECAUTIONS: Readministration) the incidence of any degree of thrombocytopenia was 5%, with an incidence of profound thrombocytopenia of 2% (<20,000 cell/μL). Factors associated with an increased risk of thrombocytopenia were a history of thrombocytopenia on previous Abciximab exposure, readministration within 30 days, and a positive HACA assay prior to the readministration.Among 14 patients who had thrombocytopenia associated with a prior exposure to Abciximab, 7 (50%) had recurrent thrombocytopenia. In 130 patients with a readministration interval of 30 days or less, 25 (19%) developed thrombocytopenia. Severe thrombocytopenia occurred in 19 of these patients. Among the 71 patients who had a positive HACA assay at baseline, 11 (15%) developed thrombocytopenia, 7 of which were severe.Allergic Reactions– There have been rare reports of allergic reactions, some of which were anaphylaxis (see WARNINGS: Allergic Reactions).Other Adverse Reactions- Table 5 shows adverse events other than bleeding and thrombocytopenia from the combined EPIC, EPILOG and CAPTURE trials which occurred in patients in the bolus plus infusion arm at an incidence of more than 0.5% higher than in those treated with placebo.The following additional adverse events from the EPIC, EPILOG and CAPTURE trials were reported by investigators for patients treated with a bolus plus infusion of Abciximab at incidences which were less than 0.5% higher than for patients in the placebo arm.Cardiovascular System: ventricular tachycardia (1.4%), pseudoaneurysm (0.8%), palpitation (0.5%), arteriovenous fistula (0.4%), incomplete AV block (0.3%), nodal arrhythmia (0.2%), complete AV block (0.1%), embolism (limb) (0.1%); thrombophlebitis (0.1%);Gastrointestinal System: dyspepsia (2.1%), diarrhea (1.1%), ileus (0.1%), gastroesophogeal reflux (0.1%);Hemic and Lymphatic System: anemia (1.3%), leukocytosis (0.5%), petechiae (0.2%);Nervous System: dizziness (2.9%), anxiety (1.7%), abnormal thinking (1.3%), agitation (0.7%), hypesthesia (0.6%), confusion (0.5%), muscle contractions (0.4%), coma (0.2%), hypertonia (0.2%), diplopia (0.1%);Respiratory System: pneumonia (0.4%), rales (0.4%), pleural effusion (0.3%), bronchitis (0.3%), bronchospasm (0.3%), pleurisy (0.2%), pulmonary embolism (0.2%), rhonchi (0.1%);Musculoskeletal System: myalgia (0.2%);Urogenital System: urinary retention (0.7%), dysuria (0.4%), abnormal renal function (0.4%), frequent micturition (0.1%), cystalgia (0.1%), urinary incontinence (0.1%), prostatitis (0.1%);Miscellaneous: pain (5.4%), sweating increased (1.0%), asthenia (0.7%), incisional pain (0.6%), pruritus (0.5%), abnormal vision (0.3%), edema (0.3%), wound (0.2%), abscess (0.2%), cellulitis (0.2%), peripheral coldness (0.2%), injection site pain (0.1%), dry mouth (0.1%), pallor (0.1%), diabetes mellitus (0.1%), hyperkalemia (0.1%), enlarged abdomen (0.1%), bullous eruption (0.1%), inflammation (0.1%), drug toxicity (0.1%).ImmunogenicityAs with all therapeutic proteins, there is a potential for immunogenicity. In the EPIC, EPILOG, and CAPTURE trials, positive HACA responses occurred in approximately 5.8% of these patients receiving a first exposure to Abciximab. No increase in hypersensitivity or allergic reactions was observed with Abciximab treatment (see WARNINGS: Allergic Reactions).In a study of readministration of Abciximab to patients (see PRECAUTIONS: Readministration) the overall rate of HACA positivity prior to the readministration was 6% and increased post-readministration to 27%. Among the 36 subjects receiving a fourth or greater Abciximab exposure, HACA positive assays were observed post-readministration in 16 subjects (44%). There were no reports of serious allergic reactions or anaphylaxis (see WARNINGS: Allergic Reactions). HACA positive status was associated with an increased risk of thrombocytopenia (see PRECAUTIONS: Thrombocytopenia).The data reflect the percentage of patients whose test results were considered positive for antibodies to Abciximab using an ELISA assay, and are highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody positivity in an assay may be influenced by several factors including sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to Abciximab with the incidence of antibodies to other products may be misleading.Warnings:Bleeding EventsAbciximab has the potential to increase the risk of bleeding events, rarely including those with a fatal outcome, particularly in the presence of anticoagulation, e.g., from heparin, other anticoagulants, or thrombolytics (see ADVERSE REACTIONS: Bleeding).The risk of major bleeds due to Abciximab therapy is increased in patients receiving thrombolytics and should be weighed against the anticipated benefits.Should serious bleeding occur that is not controllable with pressure, the infusion of Abciximab and any concomitant heparin should be stopped.Allergic Reactions (Including Anaphylaxis)Allergic reactions, some of which were anaphylaxis (sometimes fatal), have been reported rarely in patients treated with ReoPro. Patients with allergic reactions should receive appropriate treatment. Treatment of anaphylaxis should include immediate discontinuation of ReoPro administration and initiation of resuscitative measures.Overdosage:There has been no experience of overdosage in human clinical trials.Description:Abciximab, ReoPro®, is the Fab fragment of the chimeric human-murine monoclonal antibody 7E3. Abciximab binds to the glycoprotein (GP) IIb/IIIa receptor of human platelets and inhibits platelet aggregation. Abciximab also binds to the vitronectin (αvβ3) receptor found on platelets and vessel wall endothelial and smooth muscle cells.The chimeric 7E3 antibody is produced by continuous perfusion in mammalian cell culture. The 47,615 dalton Fab fragment is purified from cell culture supernatant by a series of steps involving specific viral inactivation and removal procedures, digestion with papain and column chromatography.ReoPro® is a clear, colorless, sterile, non-pyrogenic solution for intravenous (IV) use. Each single use vial contains 2 mg/mL of Abciximab in a buffered solution (pH 7.2) of 0.01 M sodium phosphate, 0.15 M sodium chloride and 0.001% polysorbate 80 in Water for Injection. No preservatives are added.
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 阿昔单抗注射剂abciximab(ReoPro injection 10mg/5m)