恩杂鲁胺胶囊enzalutamide(Xtandi 40mg Weichkapseln)
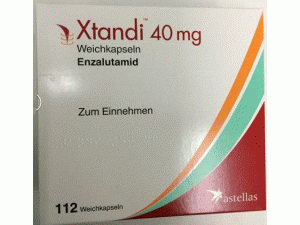 产地国家:德国
处方药:是
所属类别: 40毫克/胶囊 112胶囊/瓶/盒
包装规格: 40毫克/胶囊 112胶囊/瓶/盒
计价单位:瓶
生产厂家英文名:Milinda GmbH & Co. KG
原产地英文商品名:Xtandi 40mg/cap 112caps/box
原产地英文药品名:ESCITALOPRAM
中文参考商品译名:Xtandi 40毫克/胶囊 112胶囊/盒
中文参考药品译名:恩杂鲁胺
产地国家:德国
处方药:是
所属类别: 40毫克/胶囊 112胶囊/瓶/盒
包装规格: 40毫克/胶囊 112胶囊/瓶/盒
计价单位:瓶
生产厂家英文名:Milinda GmbH & Co. KG
原产地英文商品名:Xtandi 40mg/cap 112caps/box
原产地英文药品名:ESCITALOPRAM
中文参考商品译名:Xtandi 40毫克/胶囊 112胶囊/盒
中文参考药品译名:恩杂鲁胺
简介
部份中文恩杂鲁胺处方资料(仅供参考) 英文名:enzalutamide 商标名:XTANDI 中文名:恩杂鲁胺 生产商:安斯泰来 作用机理:恩他鲁胺是雄激素受体抑制剂,其作用于雄激素受体信号通路的不同步骤。恩扎鲁胺已被证明竞争性地抑制雄激素与雄激素受体的结合,并因此抑制雄激素受体的核移位及其与DNA的相互作用。一个主要代谢物,N-去甲基泽兰酰胺,表现出类似的体外活性Zealutut酰胺。在异种小鼠前列腺癌移植模型中,恩扎鲁胺在体外抑制了前列腺癌细胞的增殖和诱导细胞死亡,并降低了肿瘤体积。 适应症及用法:XTANDI是一种雄激素受体抑制剂,用于治疗去势抗性前列腺癌患者。 剂量与给药:XTANDI 160mg(四40mg胶囊)每日口服1次。全脂胶囊。XTANDI可以携带或不带食物。接受XTANDI的患者也应同时接受促性腺激素释放激素(GnRH)类似物或应进行双侧睾丸切除术。 剂型和强度:40mg胶囊 禁忌症:没有。 警告和注意事项:在接受XTANDI的患者中有0.4%发生癫痫发作。在易感因素患者中,2.2%的患者有癫痫发作。在治疗期间发生癫痫发作的患者永久停止XTANDI。后部可逆性脑病综合征(PRES):停止XTANDI。超敏反应:停止XTANDI。缺血性心脏病:优化心血管危险因素的管理。停止XTANDI为3-4事件。在接受XTANDI的患者中分别发生了10%和8%的跌倒和骨折。评估患者的骨折和跌倒风险,并根据既定指导方针治疗骨靶向剂。胎儿胚胎毒性:XTANDI可引起胎儿损伤和妊娠损失。建议男性具有生殖潜能的女性使用有效的避孕方法。 不良反应:XTANDI患者出现的最常见的不良反应(10%以上)为乏力/乏力、食欲减退、潮热、关节痛、头晕/眩晕、高血压、头痛和体重下降。 药物相互作用:避免强的CYP2C8抑制剂,因为它们可以增加血浆对XTANDI的暴露。如果联合用药是必要的,减少XTANDI的剂量。避免强CYP3A4诱导剂,因为它们可以降低血浆暴露于XTANDI。如果联合用药是必要的,增加XTANDI的剂量。避免CYP3A4、CYP2C9和CYP2C19底物具有狭窄的治疗指数,因为XTANDI可能降低这些药物的血浆暴露。如果XTANDI与华法林(CY2C9底物)联合使用,则进行额外的INR监测。 储存:XTANDI胶囊在20℃至25℃(68°F至77°F)干燥处储存,并保持容器密闭。允许从15°C到30°C(59°F到86°F)的偏移。英文版说明书
XTANDI(enzalutamide)capsulesXTANDI(enzalutamide) capsules is indicated for the treatment of patients with metastatic castration-resistant prostate cancer (CRPC).Important Safety InformationContraindicationsXTANDI is not indicated for women. XTANDI can cause fetal harm and potential loss of pregnancy.Warnings and PrecautionsSeizure occurred in 0.5% of patients receiving XTANDI in clinical studies. In a study of patients with predisposing factors, seizures were reported in 2.2% of patients. See section 5.1 of the Prescribing Information for the list of predisposing factors. It is unknown whether anti-epileptic medications will prevent seizures with XTANDI. Permanently discontinue XTANDI in patients who develop a seizure during treatment.Posterior Reversible Encephalopathy Syndrome (PRES) In post approval use, there have been reports of PRES in patients receiving XTANDI. PRES is a neurological disorder which can present with rapidly evolving symptoms including seizure, headache, lethargy, confusion, blindness, and other visual and neurological disturbances, with or without associated hypertension. A diagnosis of PRES requires confirmation by brain imaging, preferably MRI. Discontinue XTANDI in patients who develop PRES.Adverse ReactionsThe most common adverse reactions (≥ 10%) that occurred more commonly (≥ 2% over placebo) in the XTANDI patients from the two placebo-controlled clinical trials were asthenia/fatigue, back pain, decreased appetite, constipation, arthralgia, diarrhea, hot flush, upper respiratory tract infection, peripheral edema, dyspnea, musculoskeletal pain, weight decreased, headache, hypertension, and dizziness/vertigo. In the bicalutamide-controlled study of chemotherapy-naïve patients, the most common adverse reactions (≥ 10%) reported in XTANDI patients were asthenia/fatigue, back pain, musculoskeletal pain, hot flush, hypertension, nausea, constipation, upper respiratory tract infection, diarrhea, and weight loss.In the placebo-controlled study of patients taking XTANDI who previously received docetaxel, Grade 3 and higher adverse reactions were reported among 47% of XTANDI patients and 53% of placebo patients. Discontinuations due to adverse events were reported for 16% of XTANDI patients and 18% of placebo patients. In the placebo-controlled study of chemotherapy-naïve patients, Grade 3-4 adverse reactions were reported in 44% of XTANDI patients and 37% of placebo patients. Discontinuations due to adverse events were reported for 6% of both study groups. In the bicalutamide-controlled study of chemotherapy-naïve patients, Grade 3-4 adverse reactions were reported in 38.8% of XTANDI patients and 37.6% of bicalutamide patients. Discontinuations due to adverse events were reported for 7.6% of XTANDI patients and 6.3% of bicalutamide patients.Lab Abnormalities: In the two placebo-controlled trials, Grade 1-4 neutropenia occurred in 15% of XTANDI patients (1% Grade 3-4) and 6% of placebo patients (0.5% Grade 3-4). Grade 1-4 thrombocytopenia occurred in 6% of XTANDI patients (0.3% Grade 3-4) and 5% of placebo patients (0.5% Grade 3-4). Grade 1-4 elevations in ALT occurred in 10% of XTANDI patients (0.2% Grade 3-4) and 16% of placebo patients (0.2% Grade 3-4). Grade 1-4 elevations in bilirubin occurred in 3% of XTANDI patients (0.1% Grade 3-4) and 2% of placebo patients (no Grade 3-4).Infections: In the study of patients taking XTANDI who previously received docetaxel, 1% of XTANDI patients compared to 0.3% of placebo patients died from infections or sepsis. In the study of chemotherapy-naïve patients, 1 patient in each treatment group (0.1%) had an infection resulting in death.Falls (including fall-related injuries) occurred in 9% of XTANDI patients and 4% of placebo patients in the two placebo-controlled trials. Falls were not associated with loss of consciousness or seizure. Fall-related injuries were more severe in XTANDI patients, and included non-pathologic fractures, joint injuries, and hematomas.Hypertension occurred in 11% of XTANDI patients and 4% of placebo patients in the two placebo-controlled trials. No patients experienced hypertensive crisis. Medical history of hypertension was balanced between arms. Hypertension led to study discontinuation in < 1% of patients in each arm.Drug InteractionsEffect of Other Drugs on XTANDI Avoid strong CYP2C8 inhibitors, as they can increase the plasma exposure to XTANDI. If co-administration is necessary, reduce the dose of XTANDI.Avoid strong CYP3A4 inducers as they can decrease the plasma exposure to XTANDI. If co-administration is necessary, increase the dose of XTANDI.Effect of XTANDI on Other Drugs Avoid CYP3A4, CYP2C9, and CYP2C19 substrates with a narrow therapeutic index, as XTANDI may decrease the plasma exposures of these drugs. If XTANDI is co-administered with warfarin (CYP2C9 substrate), conduct additional INR monitoring .用药温馨提示:当您服用此药物时,需定期接受医疗专业人士的检查,以便随时针对其药效、副作用等情况进行监测。本网站所包含的信息旨在为患者提供帮助,不能代替医学建议和治疗。
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 恩杂鲁胺胶囊enzalutamide(Xtandi 40mg Weichkapseln)
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 恩杂鲁胺胶囊enzalutamide(Xtandi 40mg Weichkapseln)