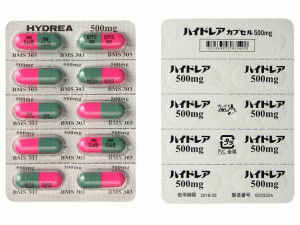
Hydrea Capsules 500mg(Hydroxycarbamide 羟基脲胶囊)说明书
 药店国别:
产地国家:日本
处 方 药:是
所属类别:500毫克/胶囊 100胶囊/盒
包装规格:500毫克/胶囊 100胶囊/盒
计价单位:盒
生产厂家中文参考译名:
百时美施贵宝
生产厂家英文名:
Bristol-Myers Squibb Corporation
原产地英文商品名:
Hydrea(ハイドレアカプセル)500mg/Capsule 100Capsules/box
原产地英文药品名:
Hydroxycarbamide
中文参考商品译名:
Hydrea(ハイドレアカプセル)500毫克/胶囊 100胶囊/盒
中文参考药品译名:
羟基脲
曾用名:
简介:
部份中文羟基脲处方资料(仅供参考)
英文名:Hydroxycarbamide
商品名:Hydrea Capsules
中文名:羟基脲胶囊
生产商:百时美施贵宝
ハイドレアカプセル500mg
药用分类名称:
抗肿瘤剂
批准日期:1992年8月
欧文商標名:
HYDREA CAPSULES 500mg
一般名:
ヒドロキシカルバミド(Hydroxycarbamide)
化学名:
ヒドロキシ尿素(Hydroxyurea)
構造式:
H2NCONHOH
分子式:
CH4N2O2
分子量:
76.05
融点:
133℃以上(分解)
性状:
羟基卡巴米德是白色至淡黄色的结晶粉末。容易溶于水和热乙醇(95),不易溶于乙醇(95),几乎不溶于二乙醚。
药效药理:
1. 抗肿瘤对可移植肿瘤的影响
(1) 除了对移植L1210白血病细胞的小鼠表现出优异的抗肿瘤作用外,还观察到各种可移植肿瘤的抗肿瘤作用,尽管是温和的。
(2) 抗肿瘤对其他耐药白血病的影响
8-阿扎鸟宁,对小鼠的抗肿瘤作用,移植L1210白血病细胞,获得对甲氨蝶酯和2-氨基-6-丁醇的耐受性。
2. 作用机制
该剂作用于细胞周期上的S期细胞,通过抑制核苷酸还原酶,将核苷酸核苷酸转化为脱氧核苷酸的酶,从而抑制DNA的合成。
适应症:
慢性骨髓性白血病、原发性血小板血症、真多血症
用法与用量:
作为羟基甲酰胺,通常口服成人每天500mg~2,000mg分为1至3次。在缓解后维持,每天口服500mg~1,000mg,分为1-2次。
此外,血液发现,症状,年龄,初始量根据体重,保持量适当增加或减少。
临床结果:
慢性骨髓性白血病的临床试验结果为91.7%(222/242),其中完全缓解为54.1%(131/242)。
包装:
胶囊 500mg: 100 胶囊 (PTP)
制造商:
百时美施贵宝有限公司
注:以上中文处方资料不够完整,使用者以原处方资料为准。
英文说明:
HYDREA CAPSULES 500mg(Hydroxycarbamide)
Brand name :HYDREA CAPSULES 500mg
Active ingredient:Hydroxycarbamide
Dosage form:
purplish red opaque/bluish green opaque hard capsule, major axis: 21.0 mm, minor axis: 7.6 mm Print on wrapping:
(Face) HYDREA 500 mg, BMS 303, (Back) ハイドレア カプセル 500 mg, ハイドレア 500mg
Effects of this medicine
This medicine suppresses the abnormal proliferation of immature bone marrow by inhibiting cellular DNA synthesis.
It is usually used for chronic myeloid leukemia, essential thrombocythemia and polycythemia vera.
Before using this medicine, be sure to tell your doctor and pharmacist
If you have previously experienced any allergic reactions (itch, rash, etc.) to any medicines.
If you have impaired renal/hepatic function, low level of white blood cells or other blood components, cold or chicken pox.
If you are pregnant, possibly pregnant or breastfeeding.
If you are taking any other medicinal products. (Some medicines may interact to enhance or diminish medicinal effects. Beware of over-the-counter medicines and dietary supplements as well as other prescription medicines.)
Dosing schedule (How to take this medicine)
Your dosing schedule prescribed by your doctor is ((to be written by a healthcare professional))
In general, for adults, take 1 to 4 capsules (500 to 2,000 mg of the active ingredient) in 1 to 3 divided doses a day. For maintenance after remission, take 1 or 2 capsules (500 to 1,000 mg) in 1 or 2 divided doses a day. The initial dose or maintenance dose may be adjusted according to the symptoms, age or body weight. Strictly follow the instructions.
If you miss a dose, skip the missed dose and follow your regular dosing schedule. You should never take two doses at one time.
If you accidentally take more than your prescribed dose, consult with your doctor or pharmacist.
Do not stop taking this medicine unless your doctor instructs you to do so.
Precautions while taking this medicine
Visit your doctor regularly to check the effects or adverse reactions of this medicine.
If you or your partner have a possibility of pregnancy, avoid pregnancy while you are taking this medicine and for a certain period after you discontinue thismedicine.
Possible adverse reactions to this medicine
The most commonly reported adverse reactions include rash/eruption, nausea/vomiting, and anemia. If any of these symptoms occur, consult with your doctor or pharmacist.
The symptoms described below are rarely seen as initial symptoms of the adverse reactions indicated in brackets. If any of these symptoms occur, stop taking this medicine and see your doctor immediately.
fever, chill, cough, nasal discharge, dizziness, bleeding tendency, continuous bleeding, bruise, dullness, easy faigability, shortness of breath, sore throat [bone marrow depression]
fever, cough, breathing difficulty [interstitial pneumonia]
lower limbs ulcer [skin ulcer]
The above symptoms do not describe all the adverse reactions to this medicine. Consult with your doctor or pharmacist if you notice any symptoms of concern other than those listed above.
Storage conditions and other information
Keep out of the reach of children. Store away from light, heat and moisture.
Discard the remainder. Do not store them. Ask your pharmacist or healthcare facility how to discard.
Internal
Revised: 1/2018
The information on this sheet is based on approvals granted by the Japanese regulatory authority. Approval details may vary by country. Medicines have adverse reactions (risks) as well as efficacies (benefits). It is important to minimize adverse reactions and maximize efficacy. To obtain a better therapeutic response, patients should understand their medication and cooperate with the treatment.
药店国别:
产地国家:日本
处 方 药:是
所属类别:500毫克/胶囊 100胶囊/盒
包装规格:500毫克/胶囊 100胶囊/盒
计价单位:盒
生产厂家中文参考译名:
百时美施贵宝
生产厂家英文名:
Bristol-Myers Squibb Corporation
原产地英文商品名:
Hydrea(ハイドレアカプセル)500mg/Capsule 100Capsules/box
原产地英文药品名:
Hydroxycarbamide
中文参考商品译名:
Hydrea(ハイドレアカプセル)500毫克/胶囊 100胶囊/盒
中文参考药品译名:
羟基脲
曾用名:
简介:
部份中文羟基脲处方资料(仅供参考)
英文名:Hydroxycarbamide
商品名:Hydrea Capsules
中文名:羟基脲胶囊
生产商:百时美施贵宝
ハイドレアカプセル500mg
药用分类名称:
抗肿瘤剂
批准日期:1992年8月
欧文商標名:
HYDREA CAPSULES 500mg
一般名:
ヒドロキシカルバミド(Hydroxycarbamide)
化学名:
ヒドロキシ尿素(Hydroxyurea)
構造式:
H2NCONHOH
分子式:
CH4N2O2
分子量:
76.05
融点:
133℃以上(分解)
性状:
羟基卡巴米德是白色至淡黄色的结晶粉末。容易溶于水和热乙醇(95),不易溶于乙醇(95),几乎不溶于二乙醚。
药效药理:
1. 抗肿瘤对可移植肿瘤的影响
(1) 除了对移植L1210白血病细胞的小鼠表现出优异的抗肿瘤作用外,还观察到各种可移植肿瘤的抗肿瘤作用,尽管是温和的。
(2) 抗肿瘤对其他耐药白血病的影响
8-阿扎鸟宁,对小鼠的抗肿瘤作用,移植L1210白血病细胞,获得对甲氨蝶酯和2-氨基-6-丁醇的耐受性。
2. 作用机制
该剂作用于细胞周期上的S期细胞,通过抑制核苷酸还原酶,将核苷酸核苷酸转化为脱氧核苷酸的酶,从而抑制DNA的合成。
适应症:
慢性骨髓性白血病、原发性血小板血症、真多血症
用法与用量:
作为羟基甲酰胺,通常口服成人每天500mg~2,000mg分为1至3次。在缓解后维持,每天口服500mg~1,000mg,分为1-2次。
此外,血液发现,症状,年龄,初始量根据体重,保持量适当增加或减少。
临床结果:
慢性骨髓性白血病的临床试验结果为91.7%(222/242),其中完全缓解为54.1%(131/242)。
包装:
胶囊 500mg: 100 胶囊 (PTP)
制造商:
百时美施贵宝有限公司
注:以上中文处方资料不够完整,使用者以原处方资料为准。
英文说明:
HYDREA CAPSULES 500mg(Hydroxycarbamide)
Brand name :HYDREA CAPSULES 500mg
Active ingredient:Hydroxycarbamide
Dosage form:
purplish red opaque/bluish green opaque hard capsule, major axis: 21.0 mm, minor axis: 7.6 mm Print on wrapping:
(Face) HYDREA 500 mg, BMS 303, (Back) ハイドレア カプセル 500 mg, ハイドレア 500mg
Effects of this medicine
This medicine suppresses the abnormal proliferation of immature bone marrow by inhibiting cellular DNA synthesis.
It is usually used for chronic myeloid leukemia, essential thrombocythemia and polycythemia vera.
Before using this medicine, be sure to tell your doctor and pharmacist
If you have previously experienced any allergic reactions (itch, rash, etc.) to any medicines.
If you have impaired renal/hepatic function, low level of white blood cells or other blood components, cold or chicken pox.
If you are pregnant, possibly pregnant or breastfeeding.
If you are taking any other medicinal products. (Some medicines may interact to enhance or diminish medicinal effects. Beware of over-the-counter medicines and dietary supplements as well as other prescription medicines.)
Dosing schedule (How to take this medicine)
Your dosing schedule prescribed by your doctor is ((to be written by a healthcare professional))
In general, for adults, take 1 to 4 capsules (500 to 2,000 mg of the active ingredient) in 1 to 3 divided doses a day. For maintenance after remission, take 1 or 2 capsules (500 to 1,000 mg) in 1 or 2 divided doses a day. The initial dose or maintenance dose may be adjusted according to the symptoms, age or body weight. Strictly follow the instructions.
If you miss a dose, skip the missed dose and follow your regular dosing schedule. You should never take two doses at one time.
If you accidentally take more than your prescribed dose, consult with your doctor or pharmacist.
Do not stop taking this medicine unless your doctor instructs you to do so.
Precautions while taking this medicine
Visit your doctor regularly to check the effects or adverse reactions of this medicine.
If you or your partner have a possibility of pregnancy, avoid pregnancy while you are taking this medicine and for a certain period after you discontinue thismedicine.
Possible adverse reactions to this medicine
The most commonly reported adverse reactions include rash/eruption, nausea/vomiting, and anemia. If any of these symptoms occur, consult with your doctor or pharmacist.
The symptoms described below are rarely seen as initial symptoms of the adverse reactions indicated in brackets. If any of these symptoms occur, stop taking this medicine and see your doctor immediately.
fever, chill, cough, nasal discharge, dizziness, bleeding tendency, continuous bleeding, bruise, dullness, easy faigability, shortness of breath, sore throat [bone marrow depression]
fever, cough, breathing difficulty [interstitial pneumonia]
lower limbs ulcer [skin ulcer]
The above symptoms do not describe all the adverse reactions to this medicine. Consult with your doctor or pharmacist if you notice any symptoms of concern other than those listed above.
Storage conditions and other information
Keep out of the reach of children. Store away from light, heat and moisture.
Discard the remainder. Do not store them. Ask your pharmacist or healthcare facility how to discard.
Internal
Revised: 1/2018
The information on this sheet is based on approvals granted by the Japanese regulatory authority. Approval details may vary by country. Medicines have adverse reactions (risks) as well as efficacies (benefits). It is important to minimize adverse reactions and maximize efficacy. To obtain a better therapeutic response, patients should understand their medication and cooperate with the treatment.
用药温馨提示:当您服用此药物时,需定期接受医疗专业人士的检查,以便随时针对其药效、副作用等情况进行监测。本网站所包含的信息旨在为患者提供帮助,不能代替医学建议和治疗。
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » Hydrea Capsules 500mg(Hydroxycarbamide 羟基脲胶囊)说明书
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » Hydrea Capsules 500mg(Hydroxycarbamide 羟基脲胶囊)说明书