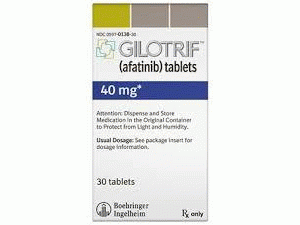
阿法替尼片(Gilotrif 50mg Tablets)
 产地国家:日本
处方药:是
所属类别: 50mg毫克/片 14片/盒
包装规格: 50mg毫克/片 14片/盒
计价单位:盒
生产厂家中文参考译名:无
生产厂家英文名:Japan Boehringer Ingelheim Pharmaceuticals
原产地英文商品名:Gilotrif tablets(ジオトリフ錠)50mg/Tablet 14Tablets/
原产地英文药品名:Afatinib Maleate
中文参考商品译名:Gilotrif片(ジオトリフ錠)50毫克/片 14片/盒(7片*2板 PTP)
中文参考药品译名:阿法替尼
曾用名:无
产地国家:日本
处方药:是
所属类别: 50mg毫克/片 14片/盒
包装规格: 50mg毫克/片 14片/盒
计价单位:盒
生产厂家中文参考译名:无
生产厂家英文名:Japan Boehringer Ingelheim Pharmaceuticals
原产地英文商品名:Gilotrif tablets(ジオトリフ錠)50mg/Tablet 14Tablets/
原产地英文药品名:Afatinib Maleate
中文参考商品译名:Gilotrif片(ジオトリフ錠)50毫克/片 14片/盒(7片*2板 PTP)
中文参考药品译名:阿法替尼
曾用名:无
简介
英文药名:Gilotrif Tablets(Afatinib Maleate) 中文药名:阿法替尼片 生产厂家:勃林格殷格翰 治疗类别名称:抗肿瘤药/酪氨酸激酶抑制剂 商标名:Giotrif Tablets 一般名:アファチニブマレイン酸塩(JAN)Afatinib Maleate(JAN) 化学名:(2E)-N-[4-(3-Chloro-4-fluoroanilino)-7-{[(3S)-oxolan-3-yl]oxy}quinazolin-6-yl]-4-(dimethylamino)but-2-enamide dimaleate 化学構造式分子式:C24H25ClFN5O3・2C4H4O4 分子量:718.08 性状:黄褐色粉末状白色。 它是易溶于水或二甲亚砜,难溶在甲醇中,微溶于乙醇(95)。分配系数登录P =4.7(PH9的或更多),日志D =3.8(pH7.4) 药效药理 1.抗肿瘤作用本剂是从H1666细胞系衍生,在体外研究中,表皮生长因子受体(的ErbB1)非小细胞肺癌,从NCI-H3255细胞系和L858R突变和与L858R突变T790M突变的EGFR基因与基因野生型衍生的非小细胞肺癌它抑制从与NCI-H1975细胞系衍生的非小细胞肺癌的生长。 2.作用机制本剂不仅EGFR和EGFR突变,不可逆地抑制属于ErbB受体家族的其他受体的酪氨酸激酶活性的HER2(-ErbB2)和ErbB4(HER4),ErbB受体家族(EGFR,HER2通过抑制均聚物和异二聚体由ErbB3的(HER3)和ErbB4形成)的活性,以阻止异常信号,抑制肿瘤细胞的生长。 适应病症 EGFR基因突变阳性的不可操作或复发性的非小细胞肺癌。 用法与用量 成年人,每天一次,每次空腹口服40毫克。所述剂量可根据病人的情况进行调整,但可增至50毫克一天1次。 包装规格片剂 20毫克:14片(7片×2)PTP30毫克:14片(7片×2)PTP40毫克:14片(7片×2)PTP50毫克:14片(7片×2)PTP 制造商:(进口出版社)日本勃林格殷格翰有限公司英文版说明
(Afatinib Maleate)New release of the world's first irreversible ErbB family inhibitor "Geotrif ® tablets" Based on the indications of EGFR gene mutation-positive inoperable or recurrent non-small cell lung cancerFirst dose of anticancer drug to be released from the tumor area pipeline of Boehringer IngelheimJapan Boehringer Ingelheim Co., Ltd. (Headquarters: Shinagawa-ku, Tokyo, President and CEO: Yoshiaki Aono, hereinafter "Nippon Boehringer Ingelheim") is an indication of the efficacy and effect of inoperable or recurrent nonsmall cell lung cancer positive for EGFR gene mutation, The oral anti-neoplastic agent/ tyrosine kinase inhibitor "Geotrif®tablet 20mg/30mg/40mg /same 50 mg" listed on the drug price on April 17th (hereinafter "Geotrif® tablet" generic name: afatinib maleic acid Salt) will be on sale from May 7th today.Lung cancer is the most deadly cancer in the world, and approximately 100,000 people are diagnosed as new lung cancer every year in Japan, and about 70,000 people died of lung cancer annually1. Of these, non-small cell lung cancer accounts for approximately 90% of cases of lung cancer.2, Geotrif® tablets bring new options for the treatment of nonsmall cell lung cancer and recurrent nonsmall cell lung cancer, which were previously considered as inoperable .Promotion of proper useNippon Boehringer Ingelheim will continue to promote proper use so that Geotrif®tablets are properly prescribed and administered to patients.Moreover, from the results of clinical trials so far, it is known that diarrhea and skin symptoms are expressed with high frequency as a side effect of Geotrif®. We will strive to provide information to healthcare professionals and patients and to promote sufficient understanding so that appropriate management can be taken against such side effects.About the outline of "Geotriff®tablets"Release date: May 7, 2014Drug Price Listing Date: April 17, 2014Approval date: January 17, 2014Product name: Geotrif ® tablet 20 mg / Geotrif ® tablet 30 mg / Geotrif ® tablet 40mg/Geotrif®tablet 50mgCommon name: afatinib maleateIndications/effects: EGFR gene mutation positive nonoperable or recurrent non small cell lung cancerDosage/administration: As an afaciniv for adults, usually 40 mg once daily is administered orally on an empty stomach. Although it may be increased or decreased according to the condition of the patient, it can be increased up to 50 mg once a day.About Geotrif®Geotrif® is the first irreversible ErbB family inhibitor in the world and covalently binds to the tyrosine kinase of the epidermal growth factor receptor (EGFR) and shows irreversible inhibitory action. Furthermore, against the tyrosine kinase of human epidermal growth factor receptor 2(HER2 (ErbB2)) and human epidermal growth factor receptor 4(ErbB4 (HER4)) belonging to the ErbB family as well as EGFR (ErbB1) It has been confirmed that sustained and selective inhibition of phosphorylation occurs.On the tumor area of ​​Boehringer IngelheimBased on outstanding scientific knowledge in the fields of respiratory diseases, cardiovascular diseases, metabolic diseases, central nervous system diseases, viral diseases, immune system diseases, Boehringer Ingelheim is an innovative anti-cancer We started a large-scale research program to develop drugs. Boehringer Ingelheim is working on the research and development of novel anticancer drugs in close collaboration with international scientific associations and multiple authoritative cancer research institutes globally.While aided by progress in science, we aim to develop a wide range of targeted therapies for various types of solid cancers and blood cancers. The tumor area pipeline of Boehringer Ingelheim continues to develop and this pipeline shows Boehringer Ingelheim's ongoing commitment to the development of the tumor area.用药温馨提示:当您服用此药物时,需定期接受医疗专业人士的检查,以便随时针对其药效、副作用等情况进行监测。本网站所包含的信息旨在为患者提供帮助,不能代替医学建议和治疗。
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 阿法替尼片(Gilotrif 50mg Tablets)
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 阿法替尼片(Gilotrif 50mg Tablets)